Glucose structure download free12/27/2023  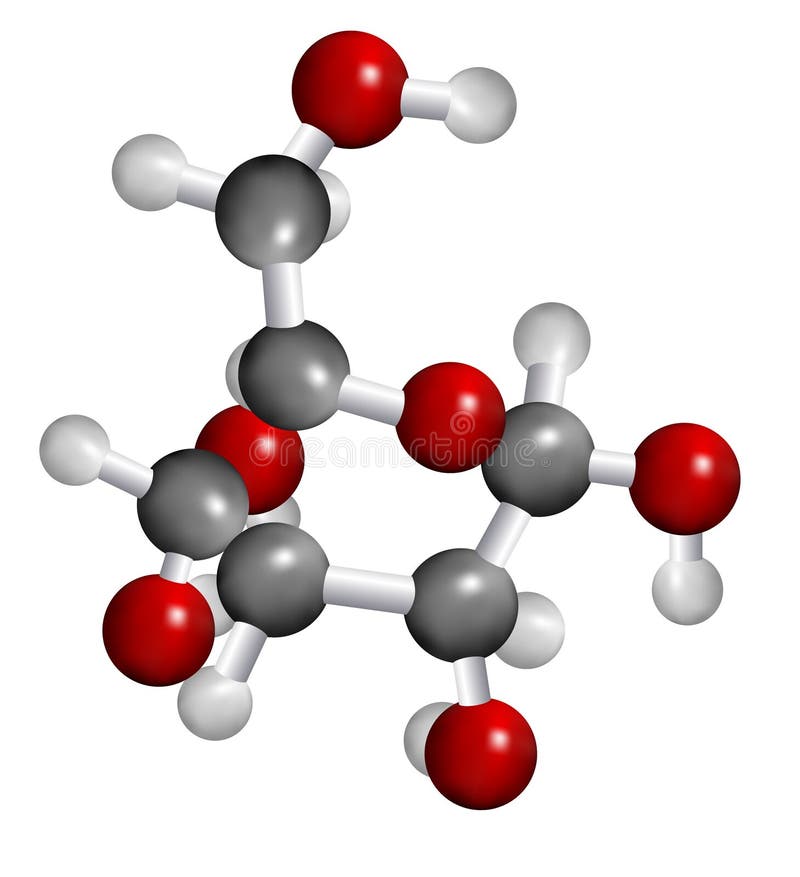
Ii) Glucose when oxidized with bromine water gives gluconic acid which when reduced with excess of HI gives n-hexanoic acid, CH 3.(CH 2) 4.COOH confirming the presence of a straight chain of six carbon atoms in glucose.ģ. This indicates that six carbon atoms in glucose are present in a straight chain. HI and phosphorus gives 2-iodohexane and n-hexane. Molecular Formula: By the usual analytical methods, the molecular formula glucose is found to be C 6H 12O 6. Commercially, it is obtained by the hydrolysis of starch which is available from relatively inexpensive source such as maize, potatoes and rice.ġ. 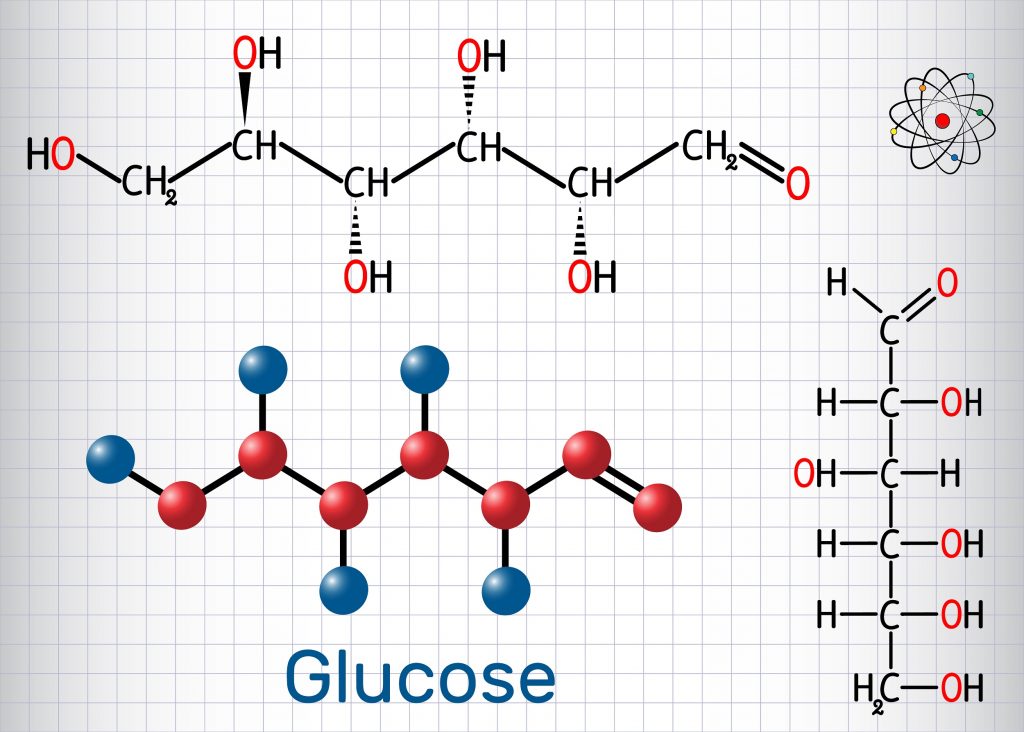
Since glucose is less soluble in ethanol, it separates out on cooling the reaction mixture. V) Glucose reacts with methanol in presence of dry HCl gas to form two isomeric glucosides. 
Iv) The existence of the two isomeric glucose and the change in specific rotation (mutarotation) is not explained by an open-chain formula. Iii) Glucose forms two isomeric pent-acetates neither of which reacts with carbonyl reagents. Ii) Glucose does not form a bisulphite and aldehyde-ammonia compound. I) Glucose does not restore Schiff's reagent colour. Objections to open-chain structure of glucose:Įven though the open chain structure of (+) glucose explains most of its reactions, it fails to explain the following facts about it. For example, the natural (-) fructose belongs to D-series, i.e., it is D(-)-fructose).ħ. 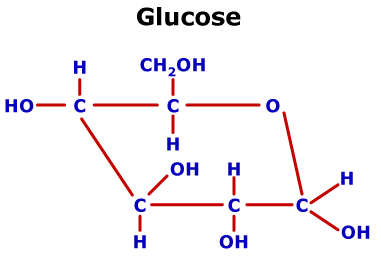
Remember that the symbols D-and L- have no relation with the specific rotation value, i.e., with (+) or (-) value. the bottom carbon atom has -OH to the left and H to the right is given L-configuration. Notations D- and L- for denoting configuration were given by Rosanoff according to this convention, any compound whose bottom asymmetric carbon atoms has the configuration similar to the configuration of dextrorotatory glyceraldehyde (drawn above, i.e. Naturally occurring glucose is the dextrorotatory glucose (+), one of the 16-stereoisomers. All these are known and correspond to the D-and L-forms of glucose, mannose, galactose, allose, glucose, idose and talose. Since the above structure possesses four asymmetric carbon atoms (shown by asterisks), it an exist in 2 4 = 16 optically active forms, i.e., eight pairs of enantiomers. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
